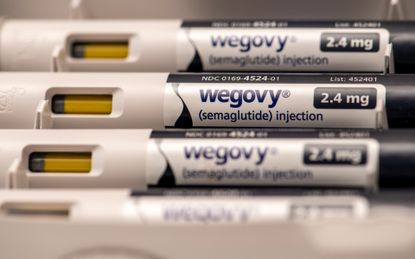
Popular weight loss drugs linked to higher risk of serious gastrointestinal problems
Researchers found that semaglutide, the active ingredient in Wegovy and Ozempic, had a higher risk for side effects like stomach paralysis.

A free daily digest of the biggest news stories of the day - and the best features from our website
Thank you for signing up to TheWeek. You will receive a verification email shortly.
There was a problem. Please refresh the page and try again.
Medicines in a popular new class of weight loss drugs including Ozempic and WeGovy may put patients at a higher risk for severe digestive problems, such as stomach paralysis, pancreatitis and bowel obstructions, according to a new study in JAMA Medical Journal. The researchers determined that the side effects were rare but still carried a higher risk than older classes of weight loss medications.
The researchers found that these side effects are rare among individual patients. For example, only about 1% of people taking Ozempic are diagnosed with stomach paralysis, but with demand for the weight loss injectables on the rise, that percentage isn't inconsequential. The study's authors warned that "even rare risks like these may amount to hundreds of thousands of new cases," CNN reported.
"When you have millions of people using these drugs, you know, a 1% risk still translates to many people who may experience these events," said Dr. Mahyar Etminan, an epidemiologist at the University of British Columbia and the lead author of the study. Still, the "study has limits" since it can't prove the drugs caused the side effects, CNN's Brenda Goodman noted.
Subscribe to The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
The study included drugs in the class known as GLP-1 inhibitors. This includes semaglutide, the active ingredient in Wegovy and Novo Nordisk's Ozempic, and liraglutide, the active ingredient in the company's older weight loss medicine Saxenda and diabetes drug Victoza, per Reuters. The researchers compared the drugs to bupropion-naltrexone, the active ingredient in weight-loss drug Contrave, which was approved in 2014.
A Novo Nordisk spokesperson noted that some of the gastrointestinal side effects in the study were already listed on the labels for its weight loss and diabetes medications. The spokesperson added that the company "stands behind the safety and efficacy of all of our GLP-1 medicines when used consistent with the product labeling and approved indications."

Continue reading for free
We hope you're enjoying The Week's refreshingly open-minded journalism.
Subscribed to The Week? Register your account with the same email as your subscription.
Sign up to our 10 Things You Need to Know Today newsletter
A free daily digest of the biggest news stories of the day - and the best features from our website
Theara Coleman is a Staff Writer for The Week. A New York native, she previously served as a contributing writer and assistant editor for Honeysuckle Magazine, where she covered racial politics and cannabis industry news. Theara graduated from Howard University and New York University, receiving her BA and MA in English Literature, respectively. She has a background in education as a former High School English teacher. She brings her passion for reading, writing, and all things nerdy to her work as a journalist.
-
 10 things you need to know today: October 7, 2023
10 things you need to know today: October 7, 2023Daily Briefing Israel 'at war' with Hamas following deadly surprise attack, Chuck Schumer leads bipartisan congressional delegation to China, and more
By Justin Klawans Published
-
 Migrants' dangerous journey across the Darién Gap
Migrants' dangerous journey across the Darién GapThe Explainer Record numbers of U.S.-bound migrants are trekking through this remote jungle. Many don’t survive the trip.
By The Week Staff Published
-
 Amazon, the 'everything store,' goes to court
Amazon, the 'everything store,' goes to courtFeature Does the retail and tech giant actually have a monopoly or is that argument a bit of a stretch?
By The Week Staff Published
-
 Consumption rooms: a legal place for illegal drugs?
Consumption rooms: a legal place for illegal drugs?Talking Point Scotland approves UK's first trial facility where users can take drugs under medical supervision
By Richard Windsor, The Week UK Published
-
 Evergreening: Big Pharma's big con
Evergreening: Big Pharma's big conThe Explainer Practice of extending patents stifles competition and can increase the cost of vital drugs to those most in need
By The Week Staff Published
-
 FDA to re-evaluate effectiveness of common nasal congestion ingredient
FDA to re-evaluate effectiveness of common nasal congestion ingredientSpeed Read
By Justin Klawans Published
-
 A flesh-eating bacteria is growing in numbers due to climate change
A flesh-eating bacteria is growing in numbers due to climate changeSpeed Read
By Devika Rao Published
-
 CDC recommends new RSV vaccine for infants under 8 months
CDC recommends new RSV vaccine for infants under 8 monthsSpeed Read
By Devika Rao Published
-
 Will Medicare drug price controls save lives?
Will Medicare drug price controls save lives?Talking Point Medicare starts negotiating lower drug prices over Big Pharma protests
By Harold Maass Published
-
 U.S. health agency advises easing federal marijuana restrictions
U.S. health agency advises easing federal marijuana restrictionsSpeed Read
By Peter Weber Published
-
 Medicare drug price negotiations start with 1st 10 drugs, pharmaceutical industry lawsuits
Medicare drug price negotiations start with 1st 10 drugs, pharmaceutical industry lawsuitsSpeed Read
By Peter Weber Published